CROSSING BIOLOGICAL BARRIERS
Active substances

Small molecules, peptides and antibodies
Formulation

Nanoparticles, microparticles, semi solids, film and patch
Innovative technologies

Laser ablation / Iontophoresis
Quantification of active substance

HPLC-UV / UPLC-MS/MS
Prodrug design
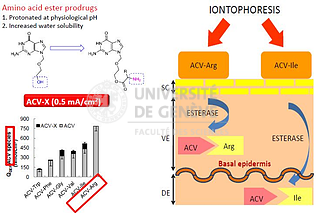
Synthesis and characterization
Visualization

Electron Microscopies
NEWS FEED
January 2024
The following paper was published:
Enhancing Oral Delivery of Biologics: A Non-Competitive and Cross-Reactive Anti-Leptin Receptor Nanofitin Demonstrates a Gut-Crossing Capacity in an Ex Vivo Porcine Intestinal Model.
Masloh S, Chevrel A, Culot M, Perrocheau A, Kalia YN, Frehel S, Gaussin R, Gosselet F, Huet S, Zeisser Labouebe M, Scapozza L. Pharmaceutics. 2024 Jan 16;16(1):116. doi: 10.3390/pharmaceutics16010116.
December 2023
The following papers were published:
Development of an ex vivo porcine skin model for the preclinical evaluation of subcutaneously injected biomacromolecules.
Gou S, Lapteva M, Brusini R, Flegeau K, Bourdon F, Kaya G, Faivre J, Kalia YN. Int J Pharm. 2023 Dec 15;648:123562. doi: 10.1016/j.ijpharm.2023.123562
Enhanced topical paromomycin delivery for cutaneous leishmaniasis treatment: Passive and iontophoretic approaches.
de Sá FAP, Andrade JFM, Miranda TC, Cunha-Filho M, Gelfuso GM, Lapteva M, Kalia YN, Gratieri T. Int J Pharm. 2023 Dec 15;648:123617. doi: 10.1016/j.ijpharm.2023.123617.
Finite sample corrections for average equivalence testing.
Boulaguiem Y, Quartier J, Lapteva M, Kalia YN, Victoria-Feser MP, Guerrier S, Couturier DL. Stat Med. 2023 Dec 19. doi: 10.1002/sim.9993.
Our communication:
Combination of thermoresponsive terbinafine nanocrystals-loaded thermo-sensitive gels and fractional ablative laser to improve ungual drug delivery.
- Rossier, Y. N. Kalia, E. Allémann.
was presented at the SFNano Annual Meeting, December 4-6, 2023, Montpellier, France.
September 2023
The following paper was published:
Polymeric micelles for cutaneous delivery of the hedgehog pathway inhibitor TAK-441: Formulation development and cutaneous biodistribution in porcine and human skin.
Darade AR, Lapteva M, Ling V, Kalia YN. Int J Pharm. 2023 Sep 25;644:123349. doi: 10.1016/j.ijpharm.2023.123349.
June 2023
We welcomed Karen Landino and Tess Vouillamoz as new PhD students in the group.
The following paper was published:
Injectable Hyaluronan-Based Thermoresponsive Hydrogels for Dermatological Applications.
Gou S, Porcello A, Allémann E, Salomon D, Micheels P, Jordan O, Kalia YN. Pharmaceutics. 2023 Jun 11;15(6):1708. doi: 10.3390/pharmaceutics15061708.
April 2023
The following paper was published:
Influence of Molecular Structure and Physicochemical Properties of Immunosuppressive Drugs on Micelle Formulation Characteristics and Cutaneous Delivery.
Quartier J, Lapteva M, Boulaguiem Y, Guerrier S, Kalia YN. Pharmaceutics. 2023 Apr 19;15(4):1278. doi: 10.3390/pharmaceutics15041278.
February 2023
Our communication:
Designing a multivalent ferritin scaffold for agonism of OX40R, a member of the tumor necrosis factor receptor super family.
- Shatz-Binder, B. Leonard, C. Blanchette, B. Kelley, Y. Kalia.
was presented at Pharmameets, February 9-11, 2023, Gaia, Portugal.
January 2023
The following paper was published:
Biodistribution of progesterone in the eye after topical ocular administration via drops or inserts.
Alambiaga-Caravaca AM, González Iglesias LG, Rodilla V, Kalia YN, López-Castellano A. Int J Pharm. 2023 Jan 5;630:122453. doi: 10.1016/j.ijpharm.2022.122453.
November 2022
We welcomed Maxime Tufeu and Xiaoxiao Wang as new PhD students in the group.
The following papers were published:
Erbium:YAG fractional laser ablation improves cutaneous delivery of pentoxifylline from different topical dosage forms.
Gou S, Del Río-Sancho S, Laubach HJ, Kalia YN. Int J Pharm. 2022 Nov 25;628:122259. doi: 10.1016/j.ijpharm.2022.122259.
Bolus delivery of palonosetron through skin by tip-loaded dissolving microneedles with short-duration iontophoresis: A potential strategy to rapidly relieve emesis associated with chemotherapy.
Kang D, Ge Q, Natabou MA, Xu W, Liu X, Xu B, Bao X, Kalia YN, Chen Y. Int J Pharm. 2022 Nov 25;628:122294. doi: 10.1016/j.ijpharm.2022.122294.
The second paper was a collaboration with the research group of Prof. Yong CHEN, a former PhD student in our group. October 6: PhD Thesis defense Aditya Darade Sep 13: PhD Thesis defense
Strategies to Improve Ocular Bioavailability and Biodistribution of Low Molecular Weight Therapeutics and Biopharmaceuticals
We welcomed Ivan Panchenko as a new PhD student in the group. The following paper was published:
Non-Invasive Iontophoretic Delivery of Cytochrome c to the Posterior Segment and Determination of Its Ocular Biodistribution.
Our communication:
Formulation and delivery strategies for sustained topical drug delivery to treat nail disorders.
was presented at the ULLA Summer School, July 2-9, 2022, Uppsala, Sweden. We welcomed Martina Ghezzi from the Department of Pharmacy and Food Sciences - University of Parma, Italy as a visiting PhD student. We welcomed Erga Syafitri as a new PhD student in the group. Our communication:
Investigation into the influence of protein structure and properties on needle-free electrically-assisted transport across the skin.
was presented at the LS2 Annual Meeting 2022: Life Sciences in the 2020s – Quantitation, Integration and Prediction, April 21-22, 2022, Campus Irchel, Zürich, Switzerland.
The following papers was published We welcomed Leqi Wang as a new PhD student in the group. The following papers was published We welcomed Alexandre Carreira Da Cruz Sousa from the Laboratory of Polymers and Materials Innovation (LabPIM) - Federal University of Ceara, Brazil as a visiting PhD student. The following papers was published We welcomed Adrián Alambiaga Caravaca from the Department of Physiology, Pharmacology and Toxicology, Universidad CEU Cardenal Herrera, Valencia, Spain as a visiting PhD student. The following papers was published The following papers was published
The influence of skin barrier impairment on the iontophoretic transport of low and high molecular weight permeants. Prof. Kalia and Dr. Lapteva were invited speakers at the “Quality & Equivalence of Topical Products & Nanotechnology Challenges” hybrid workshop (https://labhnn.com/workshop/) organised by Dr. Nathalie Hasler-Nguyen of Labhnn. Prof. Kalia’s talk was entitled “Cutaneous Biodistribution: A New Tool for Formulation Development and the Assessment of Bioequivalence of Topical Dermatological Products” and Dr. Lapteva’s talk was entitled “Opportunities of Nanotechnology for Topical Dermal Medicines”. The following papers was published The following paper, first-authored by Dr. Neeraj Thakur, was published: Jan 20: PhD Thesis defense Jan 14: PhD Thesis defense The following papers were published We welcomed Benjamin Rossier as a new PhD student in the group – his project is jointly supervised by Prof. Kalia and Prof. Eric Allémann. The following papers was published We welcomed Dr. Neeraj Thakur as a new postdoctoral scientist in the group. The following papers was published The following papers was published May 8: PhD Thesis defense The following papers were published The following papers were accepted for publication Nov 22: PhD Thesis defense The following papers were accepted for publication We had three communications at the Skin and Formulation 5th Symposium & 17th Skin Forum, September 23-24, 2019, Reims, France. We welcomed Dr. Maryam Dabbaghi from the Translational Research Center, Diamantina Institute, Faculty of Medicine, University of Queensland as a visiting student in the group. The following papers were accepted for publication We had six communications at the Controlled Release Society Annual Meeting and Exposition, July 21-24, 2019, Valencia, Spain The following paper was accepted for publication: We had communications at the 21st Annual European Pharma Congress. May 20-22, 2019, Zürich, Switzerland: Ferritin-antibody fragment conjugates: protein scaffolds to modify physicochemical and pharmacokinetic properties of biotherapeutics. and the 21st Transporter and Barrier Meeting. May 27-29, 2019, Bad Herrenalb, Karlsruhe, Germany: Investigation of apical drug transporter activity using ex vivo porcine intestine to facilitate identification of potential drug-drug-interactions at an early state of drug development. We welcomed Ms. Laeticia Pinto as a new PhD student in the group The following paper was accepted for publication The following paper was accepted for publication We had a communication at the 3rd European Conference on Pharmaceutics – Bringing science into pharmaceutical practice. March 25-26, 2019, Bologna, Italy. Investigation of drug transporter activity in ex vivo porcine intestinal mucosa We welcomed Ms. Phedra Firdaws Sahraoui as a new PhD student in the group The following paper was accepted for publication Dec 7: PhD Thesis defense The following paper was accepted for publication The following paper was accepted for publication We welcomed Mr. Fernando Sà, a visiting PhD student from the Laboratory of Food, Drug and Cosmetics, University of Brasilia, to the group. The following paper was accepted for publication We had two communication at the 12th International Conference and Workshop on Biobarriers, August 27-29, 2018, Saarbrücken, Germany. The following paper has been published from our group: We also had a communication at the 18th European Congress on Biotechnology, July 1-4, 2018, Geneva, Switzerland. Prof. Kalia was a speaker at the Proteins & Peptides: Structure, Function & Biotechnology Meeting, July 23-26, 2018, Geneva, Switzerland. June 22: PhD Thesis defense We had a communication at Phytopharm 2018 (22nd Annual Meeting), June 25-27, 2018, Horgen and ZHAW Wädenswil, Switzerland The following papers have been published from our group: We also had a communication at the EUFEPS Annual meeting, May 24-26, 2018, Athens, Greece. April 27: PhD Thesis defense April 11: PhD Thesis defense We welcomed Mr. Jonathan Faros Barros as a new PhD student in the group. We had several presentations at the 11th World Meeting on Pharmaceutics, Biopharmaceutics and Pharmaceutical Technology, March 19-22, 2018, Granada, Spain. We welcomed Mr. Aditya Darade as a new PhD student in our group. The following papers were published from our group: The following papers have been published from our group: We also had an abstract at Phytovalley, January 14-16, 2018, Seefeld, Austria.October 2022
Development of New Formulation Strategies for Targeted Drug Delivery of Low and High Molecular Weight Therapeutic Agents
September 2022
Laura Gisela González Iglesias
González Iglesias LG, Messaoudi S, Kalia YN. Pharmaceutics. 2022 Aug 31;14(9):1832. doi: 10.3390/pharmaceutics14091832.
July 2022
B. Rossier, Y. Kalia, E.Allémann
May 2022
April 2022
P. F. Sahraoui, O. Vadas, Y. N. Kalia
January 2022
Gou S, Monod M, Salomon D, Kalia YN. Pharmaceutics. 2022 Jan 24;14(2):271. doi: 10.3390/pharmaceutics14020271
Darade AR, Lapteva M, Hoffmann T, Mandler M, Schneeberger A, Kalia YN. Pharmaceutics. 2022 Jan 8;14(1):151. doi: 10.3390/pharmaceutics14010151.
October 2021
Daneluti ALM, Guerra LO, Velasco MVR, do Rosário Matos J, Baby AR, Kalia YN. Eur J Pharm Biopharm. 2021 Dec;169:113-124. doi: 10.1016/j.ejpb.2021.10.002. Epub 2021 Oct 9.September 2021
August 2021
Houriet J, Arnold YE, Pellissier L, Kalia YN, Wolfender JL. Metabolites. 2021 Aug 14;11(8):541. doi: 10.3390/metabo11080541.
Tyagi V, Serna-Jiménez CE, Kalia YN. Int J Pharm. 2021 Sep 25;607:121009. doi: 10.1016/j.ijpharm.2021.121009. Epub 2021 Aug 12.
Quartier J, Rao W, Slade S, Métral F, Lapteva M, Kalia YN. Int J Pharm. 2021 Sep 25;607:120967. doi: 10.1016/j.ijpharm.2021.120967. Epub 2021 Aug 2.
June 2021
Singhal M, Serna C, Merino V, Kalia YN. Eur J Pharm Biopharm. 2021 Sep;166:175-181. doi: 10.1016/j.ejpb.2021.06.010. Epub 2021 Jun 25.
Dahmana N, Mugnier T, Gabriel D, Favez T, Kowalczuk L, Behar-Cohen F, Gurny R, Kalia YN. Int J Pharm. 2021 Jul 15;604:120773. doi: 10.1016/j.ijpharm.2021.120773. Epub 2021 Jun 4.
May 2021
Quartier J, Lapteva M, Boulaguiem Y, Guerrier S, Kalia YN. Int J Pharm. 2021 Jul 15;604:120736. doi: 10.1016/j.ijpharm.2021.120736. Epub 2021 May 26.
Patel P, Pol A, Kalaria D, Date AA, Kalia Y, Patravale V. Eur J Pharm Biopharm. 2021 Aug;165:66-74. doi: 10.1016/j.ejpb.2021.04.026. Epub 2021 May 7
April 2021
Gratieri T, Zarhloule R, Dubey S, Kalia YN. Int J Pharm. 2021 Jun 1;602:120607. doi: 10.1016/j.ijpharm.2021.120607. Epub 2021 Apr 20.
March 2021
Int J Pharm. 2021 Mar 4;599:120445. doi: 10.1016/j.ijpharm.2021.120445
February 2021
Nanomedicine. 2021 Feb 3;33:102368. doi: 10.1016/j.nano.2021.102368
January 2021
Julie Quartier
Whitney Shatz
Pharmaceutics. 2021 Jan 11;13(1):88. doi: 10.3390/pharmaceutics13010088
Eur J Pharm Biopharm. 2021 Jan;158:379-381. doi: 10.1016/j.ejpb.2020.12.009
November 2020
Int J Pharm. 2020 Nov 15;589:119913. doi: 10.1016/j.ijpharm.2020.119913
September 2020
Biomedicines. 2020 Sep 10;8(9):340. doi: 10.3390/biomedicines8090340
June 2020
Pharmaceutics. 2020 Jun 30;12(7):608. doi: 10.3390/pharmaceutics12070608
May 2020
Si Gou
April 2020
Int J Pharm. 2020 Apr 30;580:119234. doi: 10.1016/j.ijpharm.2020.119234
Expert Opin Drug Deliv. 2020 Apr;17(4):589-602. doi: 10.1080/17425247.2020.1731470
December 2019
Dahmana N, Kowalczuk L, Gabriel D, Behar-Cohen F, Gurny R, Kalia YN.
Mol Pharm. 2019 Dec 5. doi: 10.1021/acs.molpharmaceut.9b00707
Shatz W, Blanchette C, Holder P, Kelley RF, Perozzo R, Kalia YN.
LC-GC. 2019; 37 Suppl 11 (Advances in Biopharmaceutical Analysis): 30-35.
November 2019
Vasundhara Tyagi
September 2019
Quartier J, Capony N, Lapteva M, Kalia YN.
Pharmaceutics. 2019 Sep 18;11(9). pii: E484. doi: 10.3390/pharmaceutics11090484.
J. Quartier, N. Capony, M. Lapteva, Y. N. Kalia.
J. Quartier, M. Lapteva, Y. N. Kalia. (Prize for best oral presentation)
J. Quartier, M. Lapteva, Y. N. Kalia.
August 2019
Daneluti ALM, Neto FM, Ruscinc N, Lopes I, Robles Velasco MV, Do Rosário Matos J, Baby AR, Kalia YN.
Int J Pharm. 2019 Aug 19;570:118633. doi: 10.1016/j.ijpharm.2019.118633
Tyagi V, Sancho SD, Lapteva M, Kalia YN.
Int J Pharm. 2019 Aug 12:118610. doi: 10.1016/j.ijpharm.2019.118610
July 2019
Singhal M, Merino V, Rosini M, Cavalli A, Kalia YN.
Mol Pharm. 2019 Aug 5;16(8):3460-3468. doi: 10.1021/acs.molpharmaceut.9b00252
Y. Arnold, Y. N. Kalia.
S. Gou, M. Monod, D. Salomon, Y. N. Kalia.
M. Lapteva, N. Capony, J. Quartier, Y. N. Kalia.
S. Messaoudi, L. Gonzalez-Iglesias, Y. N. Kalia.
J. Quartier, M. Lapteva, Y. N. Kalia.
P. F. Sahraoui, M. Singhal, Y. N. Kalia.
May 2019
Gou S, Del Rio-Sancho S, Singhal M, Laubach HJ, Kalia YN.
Eur J Pharm Sci. 2019 Jul 1;135:22-31. doi: 10.1016/j.ejps.2019.05.007.
W. Shatz, C. Blanchette, R. F. Kelley, R. Perozzo, Y. N. Kalia
Y. E. Arnold, Y. N. KaliaApril 2019
Kandekar SG, Singhal M, Sonaje KB, Kalia YN.
Expert Opin Drug Deliv. 2019 Apr 17. doi: 10.1080/17425247.2019.
March 2019
Arnold YE, Thorens J, Bernard S, Kalia YN.
Pharmaceutics. 2019 Mar 21;11(3). pii: E139. doi: 10.3390/pharmaceutics11030139.
Y. E. Arnold, Y. N. Kalia.February 2019
January 2019
Lapteva M, Del Río-Sancho S, Wu E, Carbonell WS, Böhler C, Kalia YN.
Sci Rep. 2019 Jan 31;9(1):1030. doi: 10.1038/s41598-018-36966-0.
Lapteva M, Mignot M, Mondon K, Möller M, Gurny R, Kalia YN.
Eur J Pharm Biopharm. 2019 Jan 12. pii: S0939-6411(18)31203-7. doi: 10.1016/j.ejpb.2019.01.008.
[Epub ahead of print]
December 2018
Somnath Kandekar
Cázares-Delgadillo J, Planard-Luong L, Gregoire S, Serna-Jiménez CE, Singhal M, Kalia YN, Merino V, Merino-Sanjuán M, Nácher A, Martínez-Gómez MA, Burnier-Yalaoui V, Barbarat P.
Pharmaceutics. 2018 Dec 7;10(4). pii: E266. doi: 10.3390/pharmaceutics10040266.
November 2018
Shatz W, Aaronson J, Yohe S, Kelley RF, Kalia YN.
Expert Opin Drug Deliv. 2018 Nov 29. doi: 10.1080/17425247.2019.1553953
September 2018
Santer V, Chen Y, Kalia YN.
Eur J Pharm Biopharm. 2018 Nov;132:157-167. doi: 10.1016/j.ejpb.2018.09.020. Epub 2018 Sep 25.
August 2018
J. Quartier, M. Lapteva, Y. N. Kalia.
J. Quartier, Y. E. Arnold, Y. N. Kalia.
July 2018
Santer V, Molliard SG, Micheels P, Río-Sancho SD, Quinodoz P, Kalia YN, Salomon D.
Dermatol Surg. 2018 Jul 9. doi: 10.1097/DSS.0000000000001609. [Epub ahead of print]
M. Lapteva, S. del Río Sancho, E. Wu, W. S. Carbonell, C. Böhler, Y. N. Kalia
June 2018
Naoual Dahmana
J. Houriet, Y. E. Arnold, C. Petit, Y. N. Kalia, J.-L. Wolfender.
May 2018
Dahmana N, Gabriel D, Gurny R, Kalia YN.
Biomed Chromatogr. 2018 May 16:e4287. doi: 10.1002/bmc.4287. [Epub ahead of print]
Yu J, Dubey S, Kalia YN.
Expert Opin Drug Deliv. 2018 Jun;15(6):559-566. doi: 10.1080/17425247.2018.1472570. Epub 2018 May 23.
S. Gou, M. Monod, D. Salomon, Y. N. Kalia.
April 2018
Verena Santer
Mayank Singhal
March 2018
M. Lapteva, S. del Río Sancho, E. Wu, W. S. Carbonell, C. Böhler, Y. N. Kalia.
N. Dahmana, R. Gurny, Y. N. Kalia.
N. Dahmana, T. Mugnier, D. Gabriel, V. Kaltsatos, T. Bertaim, Francine Behar-Cohen, R. Gurny, Y. N. Kalia.
S. Gou, S. Rio-Sancho, M. Singhal, H.-J. Laubach, Y. N. Kalia.
V. Tyagi, S. del Rio-Sancho, M. Singhal, H.-J. Laubach, Y. N. Kalia.
Y. E. Arnold, J. Thorens, S. Bernard, Y. N. Kalia.
L. G. González-Iglesias, C. Serna-Jiménez, A. Ganem-Rondero, Y. N. Kalia.February 2018
Kalaria DR, Singhal M, Patravale V, Merino V, Kalia YN.
Eur J Pharm Biopharm. 2018 Feb 22. pii: S0939-6411(17)31365-6. doi: 10.1016/j.ejpb.2018.02.031. [Epub ahead of print]
Dahmana N, Mugnier T, Gabriel D, Kaltsatos V, Bertaim T, Behar-Cohen F, Gurny R, Kalia YN.
Mol Pharm. 2018 Mar 5;15(3):1192-1202. doi: 10.1021/acs.molpharmaceut.7b01028. Epub 2018 Feb 15.
January 2018
Kandekar SG, Del Río-Sancho S, Lapteva M, Kalia YN.
Nanoscale. 2018 Jan 18;10(3):1099-1110. doi: 10.1039/c7nr07706h.
Chen Y, Kalia YN.
Int J Pharm. 2018 Jan 30;536(1):292-300. doi: 10.1016/j.ijpharm.2017.11.069. Epub 2017 Dec 2.
J. Houriet, E. Buhlmann, C. Rudigier, Y. E. Arnold, E. Kiehlmann, J. Radtke, T. Friedemann, Y. N. Kalia, S. Schröder, C. Wolfrum, J.-L. Wolfender.
